Content Testing Frequently Asked Questions
Interoperability Testing Platform (ITP) Access
Is there a cost to access the ITP Content Testing Validators?
- The Content Testing Tools are free for use by eHealth Exchange Participants and their vendors.
- If your organization does not meet either of the two criteria above, a yearly subscription can be obtained. Please send an email to testing@ehealthexchange.org for more information.
How do I get access to the Interoperability Testing Platform (ITP)?
If you are a member, participant or subscriber, you need to request a user account by sending the following information to testing@ehealthexchange.org.
- First name
- Last name
- Email address
- Phone
- Title
I have had a staff member who previously conducted our content testing leave our organization. How do I request for users to be removed from the ITP?
To have users removed, please send the user’s name and email address with the request to testing@ehealthexchange.org.
I have lost my password for the ITP, how do I reset it?
Additional information can be found in the Content Testing Tooling User Guide found here as slides #10 and #11.
What is the URL for the Content Testing Tooling component of the ITP?
Please bookmark this site https://gazellecontent.sequoiaproject.org/EVSClient/home.seam
Process
How do I gain access to the eHealth Exchange Wiki to access the content testing documentation?
- Navigate to https://ehealthexchange.org
- You will see this screen, click on the (Participant Login) in the upper right corner – see screenshot below:

- Click the Register link as shown in screenshot below:
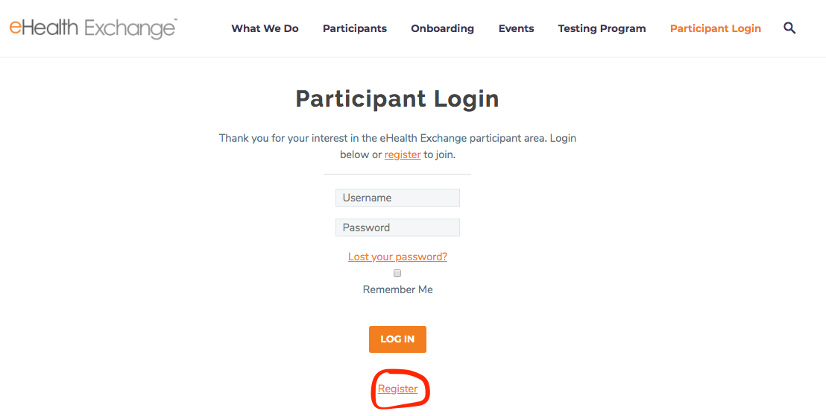
- Provide the information requested – your account will be activated immediately. Login to the wiki with your newly activated account. Make sure to navigate to the Testing wiki.
- Click on the Documentation for Content Testing Program Link as shown in the screenshot below to see the slides, testing and user guides:

I have lost my record of the permanent link references my organization submitted. How can I retrieve the permanent links previously provided?
- Your user can access the tooling at anytime to access the validation logs directly or:
- Send an email to testing@ehealthexchange.org requesting your permanent links previously submitted. These have been recorded by Sequoia and can be provided as needed.
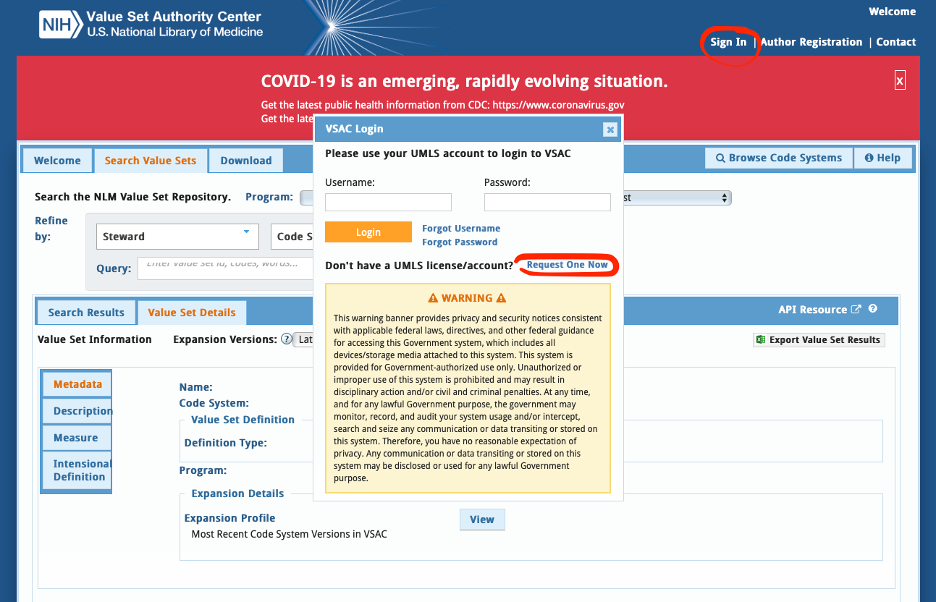
Where can we find the value sets or codes used by the content testing tooling?
- You can find all the value sets available here: https://vsac.nlm.nih.gov/. This site does require a UMLS license/account.
- To obtain a UMLS license you need to reference the screenshot below to request an account:
- Watch this video to learn how to download value sets.
Documentation
& Value Sets
How do I submit my results once testing is completed?
- Send the permanent link provided by the tooling to the email testing@ehealthexchange.org.
- Information regarding what is meant by the permanent link can be found as slide #16 within the Content Testing Tooling User Guide found here
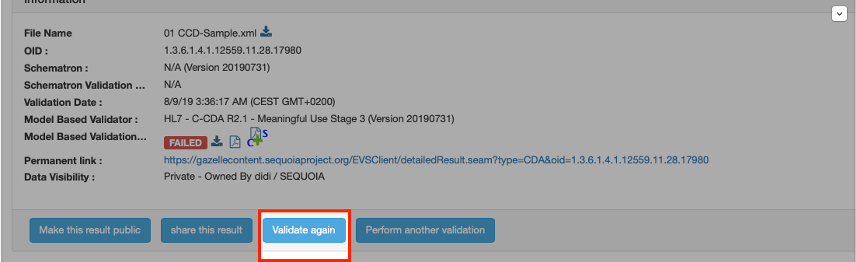
The Content Testing Tooling was updated, what do I need to do to rescore my testing previously submitted?
- Please navigate to the permanent link previously submitted and select the “Validate again” button as shown below:
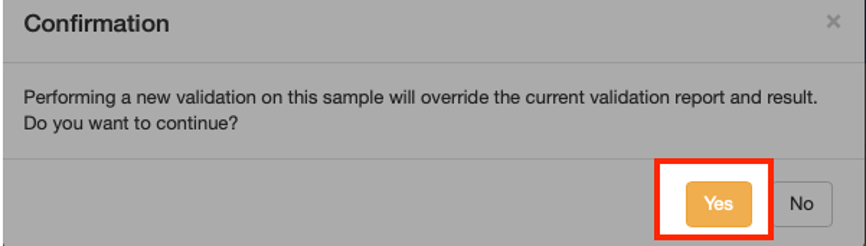
- You will be prompted to confirm that you wish to overwrite your previous report while maintaining the same permanent link for ease of tracking. Click the “Yes” button to continue as shown below:
- You will be required to select the appropriate validator for your version of document again and press the validate button as shown in the screenshots below:
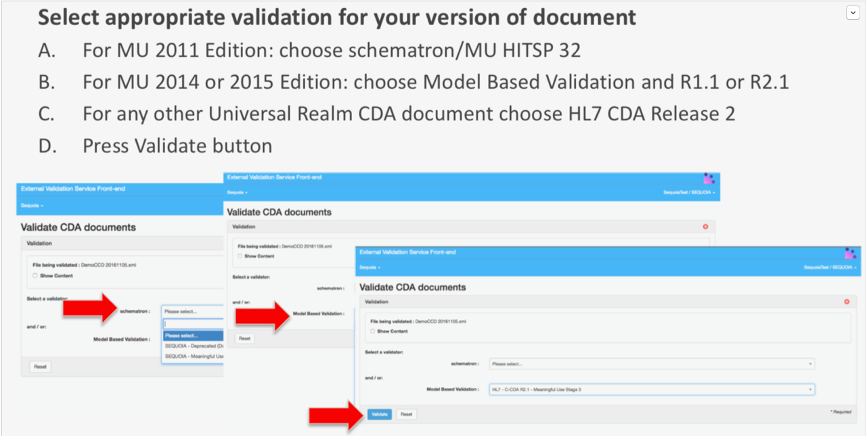
- This will overwrite your previous report while maintaining the same permanent link for ease of tracking purposes. It is recommended that you revalidate all reports previously submitted to ensure you have the correct errors identified for remediation.
We have completed our revalidation with the tooling update, do we need to officially resubmit our results, or does eHealth Exchange watch for the results via the tool?
You do not need to resubmit results unless they now pass according to the tooling revalidation. If the testing report now passes, please resend the permanent link information to the testing@ehealthexchange.org email so we can updated our records showing your passing result. We do not watch the tooling results as they are updated for pass/fail status so frequently.
How will I know which participants have not successfully passed testing?
Each organization that has not successfully passed will have their organization updated on this page https://ehealthexchange.org/participants/ under notes.


How long will we have to remediate issues found in the content testing program?
- If your organization submitted testing prior to October 1, 2019, all remediation must be completed by April 30, 2021.
- Organizations who onboard to the eHealth Exchange network after October 1, 2019 will have have 18 months from the initial testing report date to remediate any defects with their vendor or staff.
What happens if my vendor will not be able to remediate issues by the April 30, 2021 date?
Guidance was published in August 2019. Participants unable to remediate all errors and conformance issues should submit a remediation plan and/or a conformance waiver request to the email testing@ehealthexchange.org within 16 months of the date test results are submitted.
What is meant by a remediation plan?
The remediation plan should include the reason your organization cannot meet the requirement and a timeline for when the testing can will be completed.
Can I use PHI in the content testing submitted?
- No, please use ONLY test data in the document samples tested as the tooling is not hosted in a HIPAA compliant environment.
- The eHealth Exchange is working to make PHI testing available for participants in the future.
How do I report defects found within the tooling?
- Email one question/issue/defect per email to testing@ehealthexchange.org
- Provide as much information as possible including:
- Screenshots
- Testing Permanent Link with Issue
- Reference details for HL7 specification with detailed questions/issues
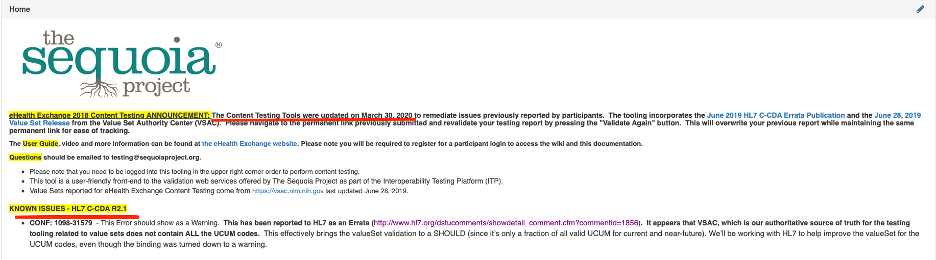
Where can I find known issues that may have already been reported for the ITP?
- The home page for the ITP Content Validators will have real time updates for any issues identified. Sequoia will coordinate with our tooling vendor to address all known issues on a quarterly basis. In addition, this page will provide information on when the validator was last updated. See screenshot below:
What is required to be remediated? Are only Errors required? What about warnings?
Only Errors MUST be fixed. Warnings are provided for your information and are typically SHOULD statements in the base specifications and associated companion guides. Your organization can choose whether to fix warnings.
I tested my content against the CDA R2 validator, why did you not accept this for formal testing?
The eHealth Exchange Coordinating Committee and Participants reached consensus in January 2017 that they would expect all clinical documents exchanged to conform to the 2011 Edition (MU1), 2014 Edition (MU2) or 2015 Edition, MU3) at a minimum. This is documented in the approved validation plan found here: https://ehealthexchange.org/testing-program/testing-references/
Other
My organization has implemented a Meaningful Use certified product. Why does the ITP show errors that the Edge Testing Tool (ETT) and/or the Standards Implementation & Testing Environment (SITE) does not.
The certification tooling maintained by ONC only tests specific document and test cases related to Meaningful Use. This ONC SITE tooling does not test all document types and value sets and does not intend to do so.
I am concerned that this initiative is taking quite a bit of time from HIE and EHR vendors, why do I need to expend the resource time to remediate issues identified?
The testing was implemented to improve the quality of clinical data exchanged to benefit clinicians and their patients. The 2018 eHealth Exchange testing aligns with the Common Clinical Data Set (CCDS). These 2015 Edition Meaningful Use requirements set the foundation for the United States Core Data for Interoperability (USCDI) that added additional data classes the industry will be required to conform to. The time spent on addressing any issues will help vendors with meeting the future USCDI requirements and should be considered time well spent towards meaningful and useful exchange of data.
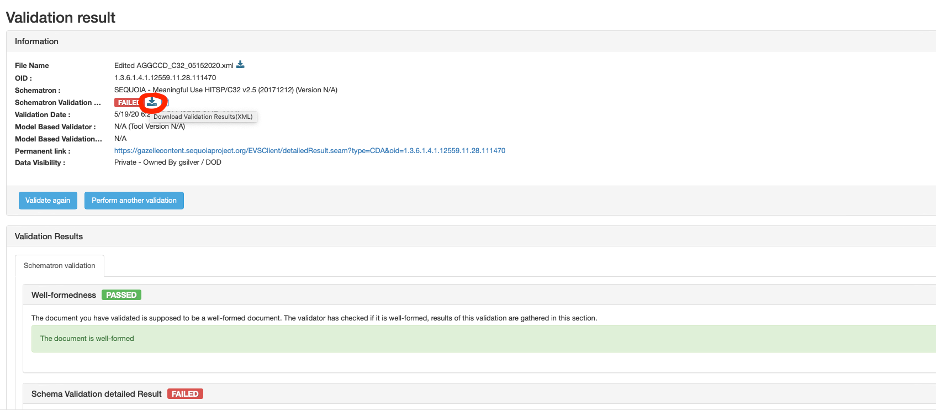
My testing report is only providing information for the first 50 errors and I have more than that according to my testing summary. How do I get information for all the errors to review?
 Click on the download validation results (XML) as shown in the screenshot below:
Click on the download validation results (XML) as shown in the screenshot below:
Are there sample C-CDA documents?
Yes, they can be found here: